Biotechnological extraction of chitin from shrimp waste for chitosan production as biostimulant in Muskmelon seeds
Abstract
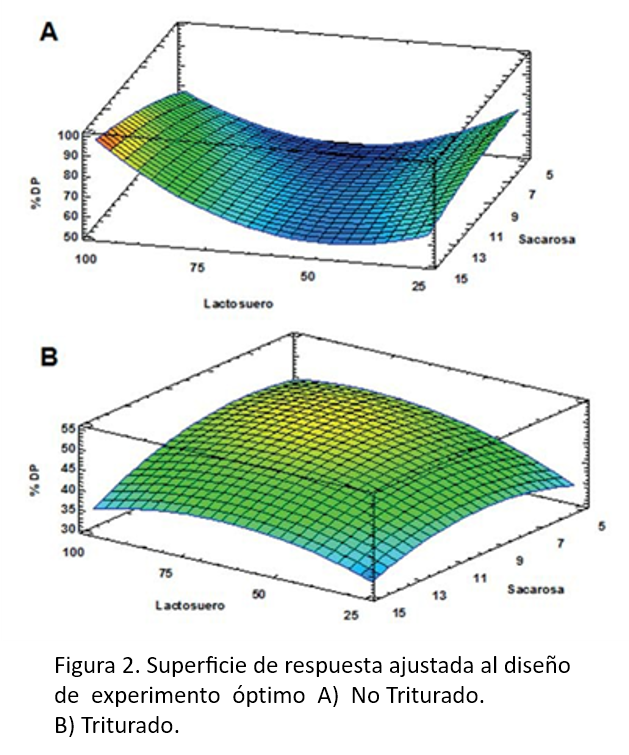
Chitin is the second most abundant biopolymer in nature after cellulose and can be obtained by various techniques. This work proposes the extraction of chitin from agroindustrial wastes and the production of chitosan as a biostimulant of Muskmelon seeds (Cucumis melo L). Chitin was isolated from the shrimp exoskeleton (Litopenaeus schmitti) by the biotechnological process of lactic acid fermentation (FAL) in stationary bioreactors, using the endemic bacteria of the shrimp cephalothorax and Lactobacillus sp. of whey. The optimization of the FAL parameters was analyzed by analysis of variance with the response surface methodology, using the percentage of deproteinization as the response variable. Chitin was purified and deacetylated (NaOH45 %) to obtain chitosan, which was evaluated by FTIR and TGA. The solubility of chitosan (> 98 %), degree of deacetylation (90 %) and molecular weight (157.68 kg / mol) were determined. Muskmelon seeds were treated with a chitosan solution, in two concentrations, 0.75 and 1 % (w/v), Q1 and Q2 respectively, evaluating 4 seed treatments (control T0, disinfection T1, disinfection + scarification T2 and scarification T3) and the effect of chitosan was compared with a commercial product. The germination percentage results for the Q1 and commercial product treatments were 100.00 and 47.21 %, and the germination rate was 1 day and 2.7 days, respectively. The T0 and T3 treatments showed better results using local soil as a substrate
Downloads
References
• Anderson, JL; Valderrama, D. 2013. Shrimp Production Review. Alliance, GA (ed.). 43 p.
• AOAC. (Official methods of analysis of AOAC International). 2005. AOAC International.
• Arbia, W; Adour, L; Amrane, A; Lounici, H. 2013. Optimization of medium composition for enhanced chitin extraction from Parapenaeus longirostris by Lactobacillus helveticus using response surface methodology. Food Hydrocolloids 31(2):392-403.
• Armenta-López, R; Guerrero, I; Huerta, S. 2002. Astaxanthin extraction from shrimp waste by lactic fermentation and enzymatic hydrolysis of the carotene protein complex. Journal of Food Science 67(3):1002-1006.
• ASTM-D446-12. 2012. Standard Specifications and Operating Instructions for Glass Capillary Kinematic Viscometers. ASTM International, WC (ed.) (D446-93). DOI: https://doi.org/10.1520/D0446-12.
• Bajaj, M; Freiberg, A; Winter, J; Xu, Y; Gallert, C. 2015. Pilot-scale chitin extraction from shrimp shell waste by deproteination and decalcification with bacterial enrichment cultures. Applied microbiology and biotechnology 99(22):9835-9846.
• Briceño, S; Suarez, J; Gonzalez, G. 2017. Solvothermal synthesis of cobalt ferrite hollow spheres with chitosan. Materials Science and Engineering: C 78:842-846.
• Bueno-Solano, C; López-Cervantes, J; Campas- Baypoli, O; Lauterio-García, R; Adan-Bante, N; Sánchez-Machado, D. 2009. Chemical and biological characteristics of protein hydrolysates from fermented shrimp by-products. Food Chemistry 112(3):671-675.
• Calderón, CS; Paz, RJ; Baudrit, JR; Ruiz-Bravo, RM. 2015. Estrategia para el desarrollo sostenible del sector social costarricense de escasos recursos económicos y la alta tecnología (en línea). Revista Digital Universitaria 16(9). Disponible en: https://bit.ly/2w14AI
• Chang, I; Prasidhi, AK; Im, J; Shin, HD; Cho, GC. 2015. Soil treatment using microbial bio- polymers for anti desertification purposes. Geoderma, 253, 39-47.
• Colina, M; Ayala, A; Rincón, D; Molina, J; Medina, J; Ynciarte, R; Vargas, J; Montilla, B. 2014. Evaluación de los procesos para la obtención química de quitina y quitosano a partir de desechos de cangrejos escala piloto e industrial. Revista Iberoamericana de Polímeros 15(1):21-43.
• COVENIN. 1979. Determinación del pH (Acidez Iónica). Alimentos Norma N° 1315:79.
• COVENIN 1986. Determinación de dureza total y calcio. Método volumétrico. Determinación de magnesio por cálculo. Agua Norma N° COVENIN 2408 – 86:10.
• COVENIN 1997. Determinación de humedad. Alimentos Norma N° COVENIN 1120-97.
• COVENIN 1999. Determinación de Cenizas. Alimentos Norma N° COVENIN 1220-99.
• Díaz Lugo, ÁA; Álvarez Castillo, RA; Méndez Martínez, JA; González Rodríguez, LV; Chocrón Castellano, MM; Guanipa Maldonado, MV. 2015. Análisis de la comunidad de peces asociada a la pesca artesanal de camarón blanco Litopenaeus schmitti, en el Golfo de Venezuela. Zootecnia Tropical 33(1):7-22.
• Francisco, FC; Simora, RMC; Nuñal, SN. 2015. Deproteination and demineralization of shrimp waste using lactic acid bacteria for the production of crude chitin and chitosan. AACL Bioflux 8(1):107-115.
• Goodrich, JD; Winter, WT. 2007. α-Chitin Nano crystals prepared from shrimp shells and their specific surface area measurement. Biomacromolecules 8(1):252-257.
• Guerra, ÁVA; Castro, LMM; Tovar, ALQ. 2013. Aprovechamiento del lactosuero como fuente de energía nutricional para minimizar el problema de contaminación ambiental. Revista de Investigación Agraria y Ambiental 4(2):55-65.
• Hofvendahl, K; Hahn–Hägerdal, B. 2000. Factors affecting the fermentative lactic acid production from renewable resources 1. Enzyme and microbial technology 26(2):87-107.
• Jung, W; Jo, G; Kuk, J; Kim, K; Park, R. 2006. Extraction of chitin from red crab shell waste by cofermentation with Lactobacillus paracasei sub sp. tolerans KCTC-3074 and Serratia marcescens FS-3. Applied microbiology and biotechnology 71(2):234-237.
• Katime, I; Cesteros, C. 2004. Química Física Macromolecular II. Servicio Editorial UPV/ EHU. (España), 350-405.
• Larez, C; Chirinos, A; Tacoronte, M; Mora, A. 2012. Chitosan oligomers as bio-stimulants to zucchini (Cucurbita pepo) seed germination. Agriculture 58(3):113-119.
• Le, TH; Nguyen, TH. 2014. Biotechnological proccess of chitin recovery from shrimp waste using Lactobacillus plantarum NCDN4. Journal of Vietnamese Environment 6(3):251-255.
• Li, Y; Sun, X; Yang, B; Ge, Y; Yi, W. 2009. Antifungal activity of chitosan on Fusarium sulphureum in relation to dry rot of potato tuber. Agricultural Sciences in China 8(5):597-604.
• Lopez-Cervantes, J; Sánchez-Machado, D; Ríos-Vázquez, N. 2006. High-performance liquid chromatography method for the simultaneous quantification of retinol, α-tocopherol, and cholesterol in shrimp waste hydrolysate. Journal of Chromatography 1105(1):135-139.
• Marcia, E; Malespín, J; Sánchez, M; Benavente, M. 2011. Estudio de la fermentación láctica para la extracción de quitina a partir de desechos de crustáceos. Nexo Revista Científica 24(1):33-42.
• Martínez González, L; Reyes Guerrero, Y; Falcón Rodríguez, A; Núñez Vázquez, M. 2015. Efecto del tratamiento a las semillas con quitosana en el crecimiento de plántulas de arroz (Oryza sativa L.) cultivar INCA LP-5 en medio salino. Cultivos Tropicales 36:143-150.
• Mogollón, JP; Rivas, W; Rivas, JG; Martínez, A. 2017. Procesos de degradación de suelos asociados a la desertificación en la península de Paraguaná, Venezuela. Ágora de Heterodoxias, 3(2), 94-110.
• Mwaheb, MA; Hussain, M; Tian, J; Zhang, X; Hamid, MI; El-Kassim, NA; Hassan, GM; Xiang, M; Liu, X. 2017. Synergetic suppression of soybean cyst nematodes by chitosan and Hirsutella minnesotensis via the assembly of the soybean rhizosphere microbial communities. Biological control 115:85-94.
• Myers, RH; Montgomery, DC; Anderson-Cook, CM. 2009. Response Surface Methodology: Process and Product Optimization using Designed Experiments. 3rd ed. Wiley (ed.). New York, 284-286
• Nascimento, WM. 2003. Muskmelon seed germination and seedling development in response to seed priming. Scientia Agricola, 60(1), 71-75.
• No, HK; Lee, SH; Park, NY; Meyers, SP. 2003. Comparison of physicochemical, binding, and antibacterial properties of chitosan prepared without and with the deproteinization process. Journal of Agricultural and Food Chemistry 51(26):7659-7663.
• Pacheco, N; Garnica-González, M; Gimeno, M; Bárzana, E; Trombotto, S; David, L; Shirai, K. 2011. Structural characterization of chitin and chitosan obtained by biological and chemical methods. Biomacromolecules 12(9):3285-3290.
• Panesar, PS; Kennedy, JF; Gandhi, DN; Bunko, K. 2007. Bioutilisation of whey for lactic acid production. Food Chemistry 105(1):1-14.
• Parada, LG; Amashta, IA; Crespín, G; Miranda, R. 2004. Caracterización de quitosano por viscosimetría capilar y valoración potenciométrica. Revista iberoamericana de polímeros 5(1):1-16.
• Peniche-Covas, C; Argüelles-Monal, W; San Román, J. 1993. A kinetic study of the thermal degradation of chitosan and a mercaptan derivative of chitosan. Polymer Degradation and Stability 39(1):21-28.
• Peña-Datoli, M; Hidalgo-Moreno, CM; González-Hernández, VA; Alcántar-González, EG; Etchevers-Barra, JD. 2016. Recubrimiento de semillas de maiz (Zea mays L.) con quitosano y alginato de sodio y su efecto en el desarrollo radical. Agrociencia 50:1091-1106.
• Plascencia-Jatomea, M; Olvera-Novoa, MA; Arredondo-Figueroa, JL; Hall, GM; Shirai, K. 2002. Feasibility of fishmeal replacement by shrimp head silage protein hydrolysate in Nile tilapia (Oreochromis niloticus L) diets. Journal of the Science of Food and Agriculture 82(7):753-759.
• Pozo, M; Azcón-Aguilar, C; Dumas-Gaudot, E; Barea, J. 1998. Chitosanase and chitinase activities in tomato roots during interactions with arbuscular mycorrhizal fungi or Phytophthora parasitica. Journal of Experimental Botany 49(327):1729-1739.
• Rao, M; Munoz, J; Stevens, W. 2000. Critical factors in chitin production by fermentation of shrimp biowaste. Applied Microbiology and Biotechnology 54(6):808-813.
• Rao, MS; Stevens, WF. 2005. Chitin production by Lactobacillus fermentation of shrimp biowaste in a drum reactor and its chemical conversion to chitosan. Journal of Chemical Technology and Biotechnology 80(9):1080-1087.
• Rinaudo, M; Milas, M; Le Dung, P. 1993. Characterization of chitosan. Influence of ionic strength and degree of acetylation on-chain expansion. International Journal of Biological Macromolecules 15(5):281-285.
• Rodríguez, AT; Ramírez, M; Falcón, A; Guridi, F; Cristo, E. 2004. Estimulación de algunas enzimas relacionadas con la defensa en plantas de arroz (Oryza sativa L.) obtenidas de semillas tratadas con quitosana. Cultivos tropicales 25(3):111-115.
• Rodríguez Beraud, M; Tampe Pérez, J; Hormazábal Vásquez, N; Araneda Durán, X; Tighe Neira, R; Cárcamo-Fincheira, P. 2017. Efecto de la escarificación y estratificación sobre la germinación in vitro de Aristotelia chilensis (Molina) Stuntz. Botánica 74(2):282-287.
• Savadogo, A; Ouattara, AC; Bassole, HI; Traore, SA. 2006. Bacteriocins and lactic acid bacteria a minireview. African journal of Biotechnology 5(9):678-683.
• Sedaghat, F; Yousefzadi, M; Toiserkani, H; Najafipour, S. 2016. Chitin from Penaeus merguiensis via microbial fermentation processing and antioxidant activity. International Journal of Biological Macromolecules 82:279-283.
• Shirai, K; Guerrero, I; Huerta, S; Saucedo, G; Castillo, A; González, RO; Hall, GM. 2001. Effect of initial glucose concentration and inoculation level of lactic acid bacteria in shrimp waste ensilation. Enzyme and Microbial Technology 28(4):446-452.
• Synowiecki, J; Al-Khateeb, NA. 2000. The recovery of protein hydrolysate during enzymatic isolation of chitin from shrimp Crangon processing discards. Food Chemistry 68(2):147-152.
• Weska, R; Moura, J; Batista, L; Rizzi, J; Pinto, L. 2007. Optimization of deacetylation in the production of chitosan from shrimp wastes: Use of response surface methodology. Journal of Food Engineering 80(3):749-753.
• Xu, Y; Gallert, C; Winter, J. 2008. Chitin purification from shrimp wastes by microbial deproteination and decalcification. Applied Microbiology and Biotechnology 79(4):687-697.
• Zawadzki, J; Kaczmarek, H. 2010. Thermal treatment of chitosan in various conditions. Carbohydrate Polymers 80(2):394-400.
• Ziani, K; Ursúa, B; Maté, JI. 2010. Application of bioactive coatings based on chitosan for artichoke seed protection. Crop Protection. 29(8):853-859.